Let’s discuss the question: how many atoms are in 123 g of calcium. We summarize all relevant answers in section Q&A of website Myyachtguardian.com in category: Blog MMO. See more related questions in the comments below.

Table of Contents
How many atoms are in 125 g of calcium?
There are 18.81875×1023 18.81875 × 10 23 atoms of calcium in a sample of 125 grams of calcium.
How many atoms are in g of calcium?
Explanation: The Periodic Table tells us that 6.022×1023 individual calcium atoms have a mass of 40.1⋅g .
How To Convert Grams to Atoms – THE EASY WAY!
Images related to the topicHow To Convert Grams to Atoms – THE EASY WAY!

How many atoms are in 120 grams of calcium?
Expert-verified answer
=5.179 ×10^23 atoms.
How many atoms does a calcium have?
| atomic number | 20 |
|---|---|
| atomic weight | 40.078 |
| melting point | 842 °C (1,548 °F) |
| boiling point | 1,484 °C (2,703 °F) |
| specific gravity | 1.55 (20 °C, or 68 °F) |
How many atoms are in 131 g of calcium?
Answer. There are 3.275 moles of calcium in 131 grams as relative atomic mass of calcium is 40. Therefore there are 1.972 x 10 24 atoms.
How many atoms are in 10g of calcium?
Answer. =0.25 gram atoms.
How many calcium atoms are in 2 grams of calcium?
= 3.011×10^22 atom will be contained in 2 grams .
How many atoms are there in 20 grams of calcium?
Answer: 3.011 x 10^23.
How do you convert grams to atoms?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro’s number: 6.02 x 10^23.
How many atoms are there in 60g of calcium?
Answer. The moles present in 60 g of calcium are 1.5 moles. Hope it helped you.
How many atoms are in calcium carbonate?
Calcium carbonate has the formula CaCO3. This means that one molecule of Calcium Carbonate has 5 atoms in total – one calcium atom, one carbon atom, and 3 oxygen atoms.
How many atoms are in 3.75 GC?
There are 2.26 x 1024 carbon atoms in 3.75 moles of carbon.
How many Atoms are in 6 g of carbon?
Images related to the topicHow many Atoms are in 6 g of carbon?

How many atoms are there in 25 grams of calcium?
The molecular weight of calcium carbonate is 100 g per mole. 25 g of calcium carbonate corresponds to 0.25mole. 1 molecule of calcium carbonate contains three oxygen atoms. Thus, 0.25 mole of calcium carbonate will contain 0.75 moles of oxygen atoms which is equal to 0.75×6.02×10^23=4.51×10^23 oxygen atoms.
How many atoms are in 3 moles of calcium?
1 mole of any material contains the Avogadro number of particles, that is, 6.023×1023 particles. Hence, by ratio and proportion, 3.2 moles of calcium will contain 3.2×6.023×1023 Ca particles or atoms, which is 1.927×1024 Ca atoms.
How many atoms are in 100g of calcium?
= 5/2 × 6.22 ×10^23 atoms.
How many grams are in 6.02 e23 atoms of calcium?
Calcium’s atomic mass is 40.08amu or 40.08 grams per mole. One mole of calcium contains 6.02 x 10 to the 23rd power(6.02e23) atoms. Take 0.6 grams and divide by 40.08 grams (0.0150) and multiply this factor by 6.02e23.
How many atoms are in a 24 g sample of carbon?
There are 1.2×10^24 atoms in 24 grams of carbon. Because Carbon’s Molar Mass is about 12 grams (the actual number is 12.011, which isn’t a large enough difference to have an especially relevant effect), this means we have 2 moles of Carbon. In one mole of atoms, there are 6.02*10^23 atoms.
How many atoms of calcium are in a 40.08 gram sample?
40.08 grams of calcium is one mole (see Periodic Table), and one mole is 6.02 x 1023 atoms. 2. One mole of anything is 6.02 x 1023, so it is 6.02 x 1023 Mg atoms.
How many atoms are present in 10 gms of calcium carbonate?
One molecule of calcuim carbonate contains 5 atoms.
How many grams atoms and atoms are there in 10 grams of calcium?
(1). 10 gm of calcium = 10 * 6.022 * 1023 / 40 = 1 . (2). Ca weigh =(40g)6.022×1023×6.022×1022=4g.
How many atoms are there in 6g of carbon?
Answer: Answer: Since 12 grams of carbon contains 1 mole of its atoms, 6 grams of it contains half a mole, that is, 3.01 x 10^23 atoms of it.
How many atoms are present in 40mg of calcium?
Chemistry IB P. 1.15 How many atoms are present in 2 grams of calcium? (GAW of Ca is 40g) Solution 40 orams of ca has 6022 x 1023 atoms.
Calculate the Mass of a Single Atom or Molecule
Images related to the topicCalculate the Mass of a Single Atom or Molecule
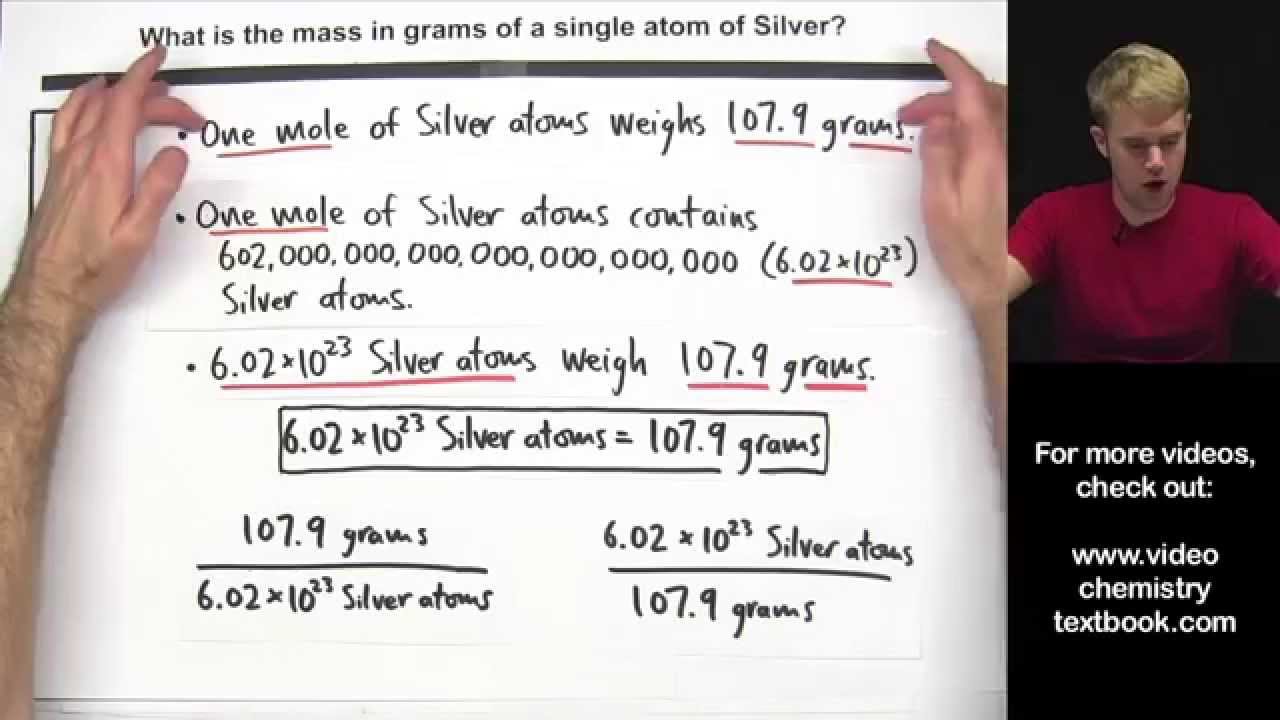
How many oxygen atoms are in 120 grams of nitric acid?
= 3.442 x 10^24 atoms of oxygen.
How many atoms does CL have?
Chlorine molecules are composed of two atoms (Cl2).
Related searches
- how many atoms are in 1 gram of calcium
- how many atoms of calcium are present in 58 2 g of calcium
- how many atoms are in 50g of calcium
- how many atoms are in 175 g of calcium
- how many moles of atoms are in 500 mg of calcium
- how many atoms are in 119 g of calcium
- how many atoms in 143 g of calcium
- how many atoms are in 117 g of calcium
- how many atoms are in 121 g of calcium
- how many atoms are in 171 g of calcium
- how many atoms are in 135 g of calcium
- how many moles are in 20 grams of calcium
Information related to the topic how many atoms are in 123 g of calcium
Here are the search results of the thread how many atoms are in 123 g of calcium from Bing. You can read more if you want.
You have just come across an article on the topic how many atoms are in 123 g of calcium. If you found this article useful, please share it. Thank you very much.

